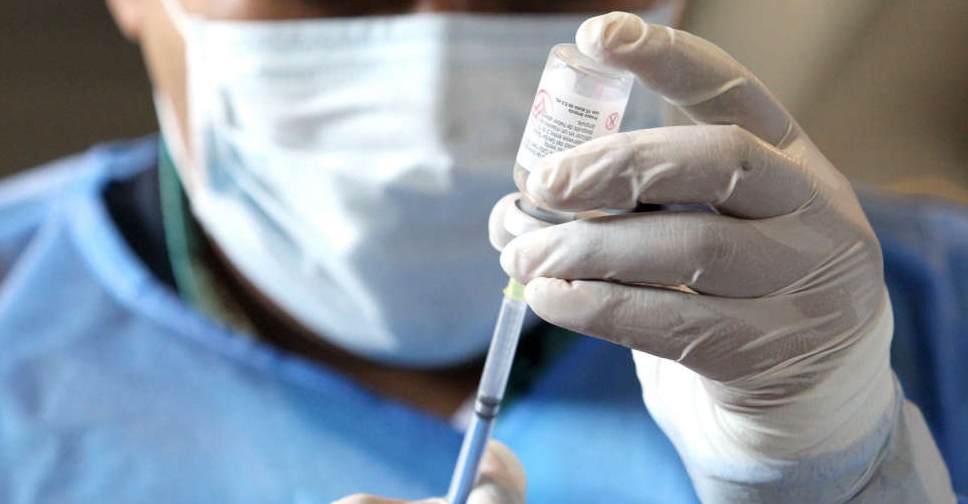
The World Health Organization (WHO) on Thursday issued an emergency use listing for the single-dose COVID-19 vaccine from China-based CanSino Biologics.
The vaccine, Convidecia, is the eleventh shot against the coronavirus to get clearance from the global health agency, whose advisory group recommended its use in people of age 18 years and above.
It was found to have 64 per cent efficacy against symptomatic disease and 92 per cent against severe COVID-19, the agency said.
Other vaccines that have similar clearance from the agency include those made by Pfizer and BioNTech, Moderna, AstraZeneca, Johnson & Johnson and Novavax.




 Trump vows no more attacks by Israel on Iran gas field after it 'violently lashed out'
Trump vows no more attacks by Israel on Iran gas field after it 'violently lashed out'
 Saudi Arabia trust with Iran hard to 're-establish', foreign minister says
Saudi Arabia trust with Iran hard to 're-establish', foreign minister says
 Kuwait Petroleum Corporation contains fire after drone hits refinery
Kuwait Petroleum Corporation contains fire after drone hits refinery
 QatarEnergy says several LNG facilities targeted by missiles, fires contained
QatarEnergy says several LNG facilities targeted by missiles, fires contained
 Kuwait foils terror plot targeting vital facilities
Kuwait foils terror plot targeting vital facilities







